Flow Chemistry in Drug Discovery
The past decade has seen a dramatic shift in the acceptance and implementation of continuous flow chemistry in drug discovery, development, delivery, and production. Improving reaction efficiencies, saving on lab resources (space, time, and costs), improving safety, and accessing new chemistries not possible with traditional batch methods are just some of the reasons for the adoption of the range of applications this technology offers.

Continuous Flow Manufacturing in Pharmaceutical Development
With more and more chemists understanding the application of flow chemistry and continuous flow techniques and the benefits it provides, combined with external influences such as the FDAs approval for continuous manufacturing, pharmaceutical chemists, and senior management at all stages of the drug lifecycle are looking to continuous flow reactor to speed up and improve the drug discovery and development process.
While chemists at each stage of the drug discovery and development process will see specific benefits when implementing continuous flow techniques, the pharmaceutical industry’s desire to ultimately move towards the continuous manufacturing of future drugs means all stages of the drug lifecycle need to be aware of – and potentially implementing – continuous flow.
Our focus here is on the impact and implementation of using flow chemistry in drug discovery, the key drivers in the drug development process and to highlight how automation is helping early hit-to-lead and lead-generation programs.
Laboratory automation removes the repetitive processes chemists are often faced with, enables reactions to be reproduced with accuracy and generally allows for increased efficiency. It also frees chemists to focus on other tasks, whether that’s catching up on publications, designing the next reaction or attending that next meeting.
This article looks to explore the benefits of laboratory automation including some key drivers for its use in both batch and flow chemistry applications.
The Medicinal Chemistry and Drug Discovery Process
Pursuing new classes of compounds which can be used as new medicines for treatment of diseases is not a simple task. Researchers often create new active compounds from scratch, through a laborious process involving synthesizing often complex organic chemistry routes and testing thousands of compounds aiming to find those suitable to be tested in human beings.1
In this context, the role of Medicinal Chemistry in drug development is an interdisciplinary science at the interface of chemical biology, pharmacology, and medicine playing a fundamental role to define novel therapeutic approaches and discover new drugs. One of the major purposes of medicinal chemistry is the design and synthesis of new lead compounds for druggable targets.2,3
In these early stages of drug discovery, chemists aim to design and synthesize molecules with specific properties in mind. The molecules should interact with specific targets, as example, proteins suspected to cause the disease. The inhibition or activation of that specific protein potentially help to treat the disease. A good drug candidate should have specific physical-chemical properties, like suitable pharmacokinetics and safety.1
Pharmaceutical companies invest large amounts of money and time into launching a new drug to market, which can take on average more than a decade of research and with an estimated cost of hundreds of millions of dollars.4 This process involves the synthesis and testing of tens of thousands of exploratory compounds to disclose one new drug. Figure 1 shows the average number of compounds required at each step of bringing a new drug to market over all stages.
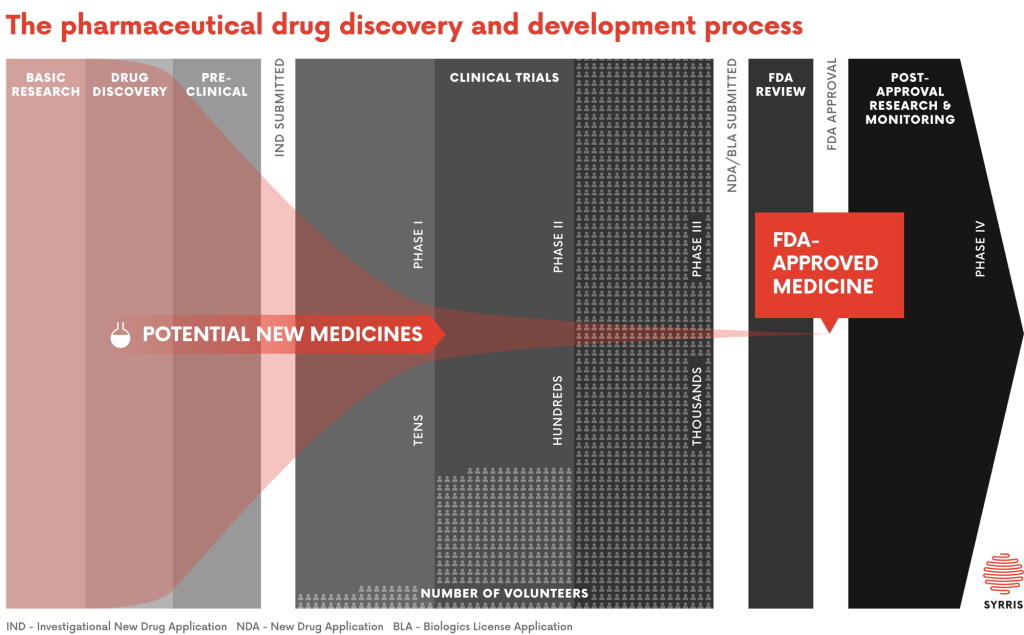
The Drug Discovery Cycle – Synthesis of Pharmaceutical Ingredients
Medicinal chemistry is a complex process that relies on iterative learning cycles composed of molecular design, chemical synthesis, testing, and structure−activity/structure−property relationship (SAR/ SPR) analysis.2 This is true for both the hit- and lead generation phases of the drug discovery process.
The selection of which series of compounds to pursue can have a huge impact on the success rates of the drug discovery and drug development process. In the first instance identifying the starting point, or “Hit” involves screening large numbers of compounds. Once the “Hits” are found, usually showing some efficacy against the target the iterative cycle begins to find a “lead” which shows improvements e.g. in binding or pharmacokinetics. These “leads” are then optimized, once again using the described iterative cycle focusing on selectivity, ADMET (absorption, distribution, metabolism, excretion, and toxicity) etc.
The decision/learning cycle is critical in identifying potential drug candidates. The need to increase the cycle rate of this decision process is a key factor in more efficient drug discovery. Faster iterative cycles enable faster decisions and generate leads faster.
Figure 2 shows the design, make, test cycle with some of evaluation of this process highlighted.
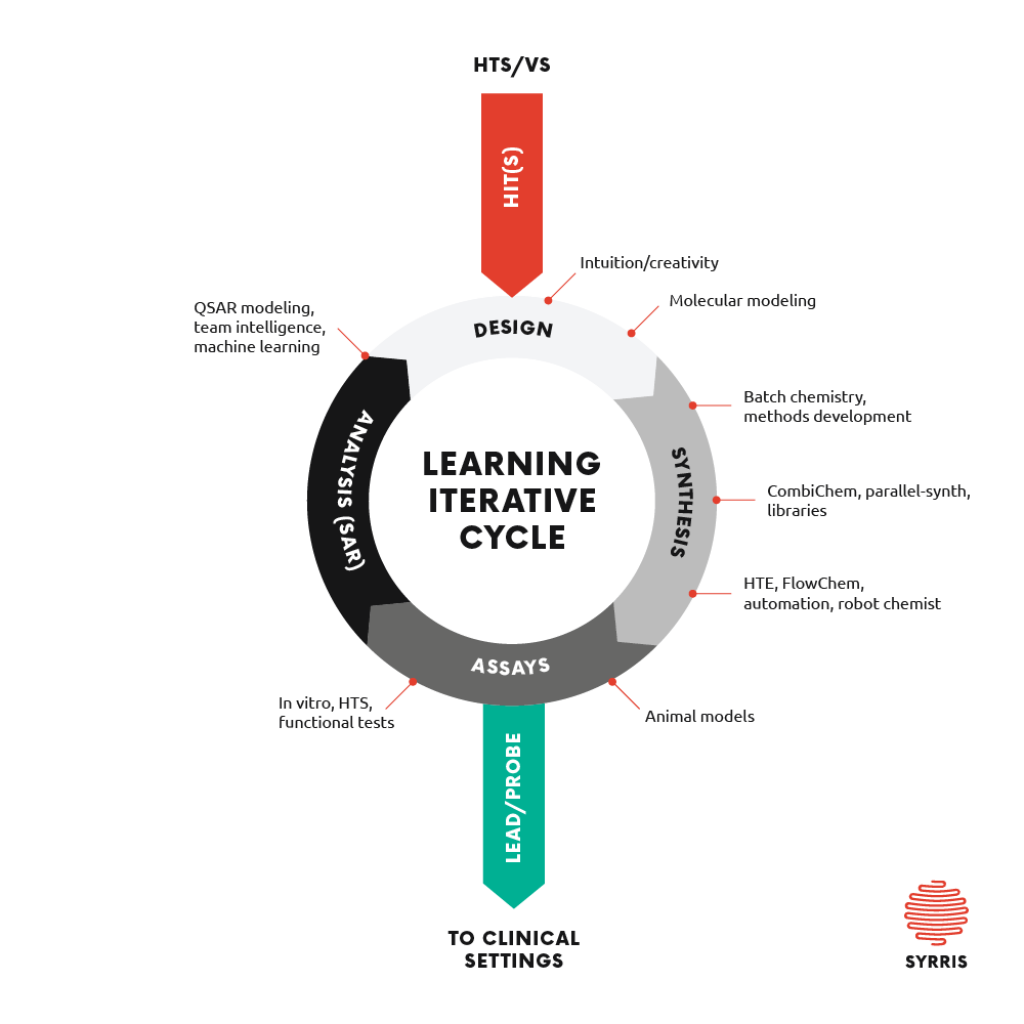
Development of Automation in the Drug Discovery Process
Until the 1980s the information regarding biological targets, their mechanisms and therefore their potential therapeutic applications was limited.5
Because of these older approaches drug discovery relied on the intuitive design of compounds, synthesized on gram quantities for animal model testing due to lack of the in-vitro screening methods available at the time. This often delivering only a few compounds per week resulting in long timelines. The development of high-throughput screening (HTS) and in-vitro models over the years enabled testing of a greater numbers of compounds in shorter time frames. This in turn led to the requirement and development of methods to synthesis compounds in larger numbers. Techniques such as combinatorial chemistry and parallel chemistry methods using automation to create huge numbers of compounds led in turn to speeding up the learning cycle.
The development of these automated techniques using computation modelling, high throughput experimentation and high throughput screening with in-vitro methods have helped accelerate the early drug discovery process generating large numbers of compounds to evaluate.
From a medicinal chemist’s view point the synthesis of diverse compounds to screen is a key part of the drug discovery process. Chemists are constantly looking for alternative solutions to solve limitations of chemical synthesis, to increase the tools in their toolbox. The adoption of enabling chemical technologies as automation and flow systems have enabled chemists to shorten iterative learning cycles through early discovery to production and to the point of care.
The International Union of Pure and Applied Chemistry (IUPAC) in 2019 named flow chemistry among the top ten emerging technologies in chemistry6 and the US regulatory agency Food and Drug Administration (FDA) declared continuous manufacturing (CM) as one of the most important tool in the modernization of the pharmaceutical industry.7
The flow chemistry (or continuous flow) processes facilitates aspects as automation, parallelization, and integration of chemical synthesis with purification and analysis, thus improved parameters that are essential to assure the constant and rapid supply of pure compounds ready for testing as well as to improve reproducibility and costs if compared with manual, serial compound synthesis.3 The range of available automated flow platforms such as the Asia Flow Chemistry system enables chemists to access a complete range of applications from continuous flow photochemistry and flow electrochemistry to develop cleaner, green chemistry.
Flow Chemistry in Drug Discovery
The flow chemistry processes facilitate aspects such as automation, parallelization, and integration of chemical synthesis with purification and analysis, thus improving the parameters essential in delivering the constant and rapid supply of pure compounds ready for testing as well as to improve reproducibility and costs if compared with manual, serial compound synthesis.8,9
The impact of Flow chemistry has seen this enabling technology being applied to a growing number of applications over the years in modern laboratories. The development of fully automated flow-based approaches has helped in the acceleration of its adoption.
There are many benefits from using flow chemistry in modern chemical synthesis (What are the benefits of flow chemistry), such as increased control of reaction parameters, improved selectivity and yields and increased reaction rates which are key in the organic chemistry performed to develop drug molecules. There are many benefits that apply specifically to the application of flow chemistry to Drug Discovery programs,
- Access to challenging or restrictive chemistry limited with traditional batch techniques
- Improved safety profiles with the creation of reactive intermediates and hazardous reagents in-situ
- Easy access to scale-up where reactions can be optimized on small scale and scaled with no re-optimization
- Efficient serial library generation with the rapid exploration of diverse chemical space
- Ability to perform multi-step chemistry using a modular approach that allows direct synthesis of complex reactions
- Easier access to reagentless chemistry such as continuous flow photochemistry and flow electrochemistry.
- Greener and more efficient processes saving money and the environment.
You can learn more about flow chemistry with our flow chemistry learning series and flow chemistry application pages.
The drug discovery process has undergone a series of continuous improvements over the past two decades in the way it carries out early drug discovery programs. The increased demand on all disciplines involved, especially chemists has seen a drive towards implementing new enabling technologies to reduce the time and cost in bringing novel drugs to market. One of these technologies, flow chemistry, has made an impact in most areas of this process, in drug discovery, development, delivery and production. The development of automated techniques and the integration of synthesis with purification and analysis has greatly increased the speed with which novel compounds can be delivered and has helped reduce the time cycles required in the early stages of drug discovery.
For further examples of how using flow chemistry can impact the drug discovery and development process this customer story outlines how Gideon Richter has implemented this technology with a continuous flow approach.
References
(1) Novartis. Discovery The art of drug design in a technological age https://www.novartis.com/stories/art-drug-design-technological-age (accessed Mar 14, 2022).
(2) Cerra, B.; Carotti, A.; Passeri, D.; Sardella, R.; Moroni, G.; Di Michele, A.; Macchiarulo, A.; Pellicciari, R.; Gioiello, A. Exploiting Chemical Toolboxes for the Expedited Generation of Tetracyclic Quinolines as a Novel Class of PXR Agonists. ACS Med. Chem. Lett. 2019, 10 (4), 677–681. https://doi.org/10.1021/acsmedchemlett.8b00459.
(3) Gioiello, A.; Piccinno, A.; Lozza, A. M.; Cerra, B. The Medicinal Chemistry in the Era of Machines and Automation: Recent Advances in Continuous Flow Technology. J. Med. Chem. 2020, 63 (13), 6624–6647. https://doi.org/10.1021/acs.jmedchem.9b01956.
(4) López, E.; Alcazar, J. Flow Chemistry in Drug Discovery: Challenges and Opportunities. In Flow Chemistry in Drug Discovery; Springer International Publishing: Berlin, Heidelberg, 2021; pp 1–22. https://doi.org/10.1007/978-3-030-85592-5.
(5) Lombardino, J. G. Loew, J. A, The role of the medicinal chemistry in drug discovery – then and now. Nat. Rev. Drug Dicovery, 2004, 3, 853 – 862.
(6) Gomollón-Bel, F. Ten Chemical Innovations That Will Change Our World: IUPAC Identifies Emerging Technologies in Chemistry with Potential to Make Our Planet More Sustainable. Chem. Int. 2019, 41 (2), 12–17. https://doi.org/10.1515/ci-2019-0203.
(7) Gottlieb, S.; Woodcock, J. FDA statement on FDA’s modern approach to advanced pharmaceutical manufacturing https://www.fda.gov/news-events/press-announcements/fda-statement-fdas-modern-approach-advanced-pharmaceutical-manufacturing (accessed Mar 15, 2022).
(8) Schneider, g. Automating drug discovery. Nat. Rev. Drug Discovery, 2018, 17, 97 – 113
(9) Trobe, M.; Burke, M. D. The molecular industrial revolution: automating synthesis of small molecules.Angew. Chem., Int. Ed. 2018, 57, 4192 – 4214.
How Syrris can help?
Syrris is the longest established flow chemistry equipment supplier and has been producing premium flow chemistry systems since 2001. Starting with the first of its kind automated flow chemistry system aimed at the Pharmaceutical industry, Syrris AFRICA set the bench-mark for the development of commercial flow chemistry systems and the adoption of flow chemistry in industry. Syrris Flow Chemistry products are now in their 4th iteration showing our commitment to the growing requirements of chemists in continued development of flow chemistry systems that met industry and academic research needs.
The Syrris Asia Flow Chemistry system is an award-winning family of flow products from Syrris. Designed by chemists for chemists, it enables the widest variety of chemical reactions and ultimate ease of use. Combined with Asia Manager PC Control Software an almost unlimited combination of flow chemistry applications can be automated.

Contact our team of specialists to find out how the Syrris Asia Flow Chemistry System could help your chemistry applications.
