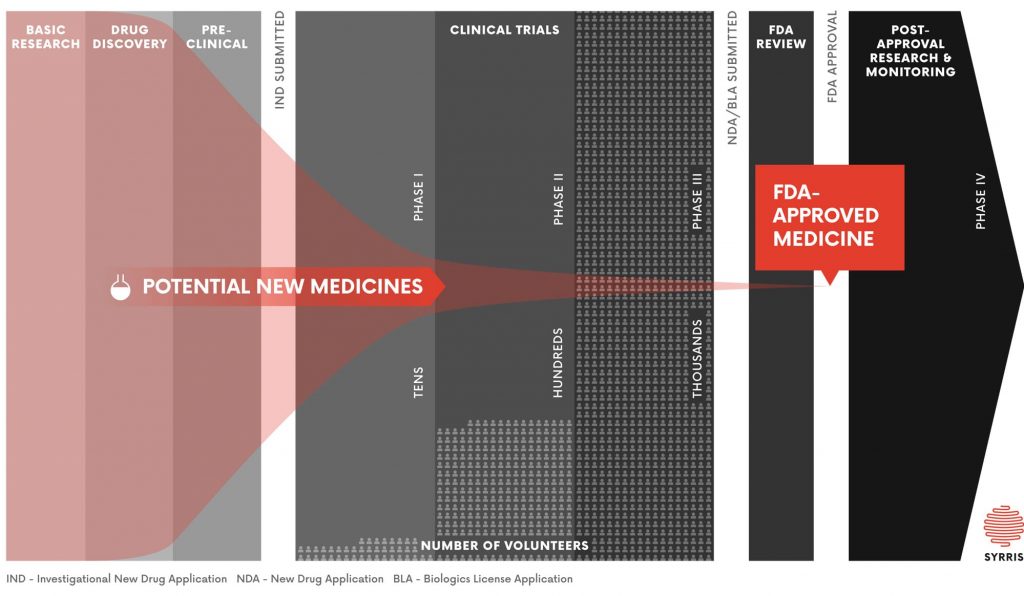
The drug discovery process
Drug discovery typically begins with target identification – selecting a biochemical mechanism involved in a disease condition. Up to 10,000 drug candidates are then put through a rigorous screening process to establish their interaction with the drug target. Once initial “hits” (drugs with activity against the target) are identified, a focused library of similar compounds will be synthesized to optimize interactions with the drug target; this is the process of library synthesis.
Library synthesis of drug candidates
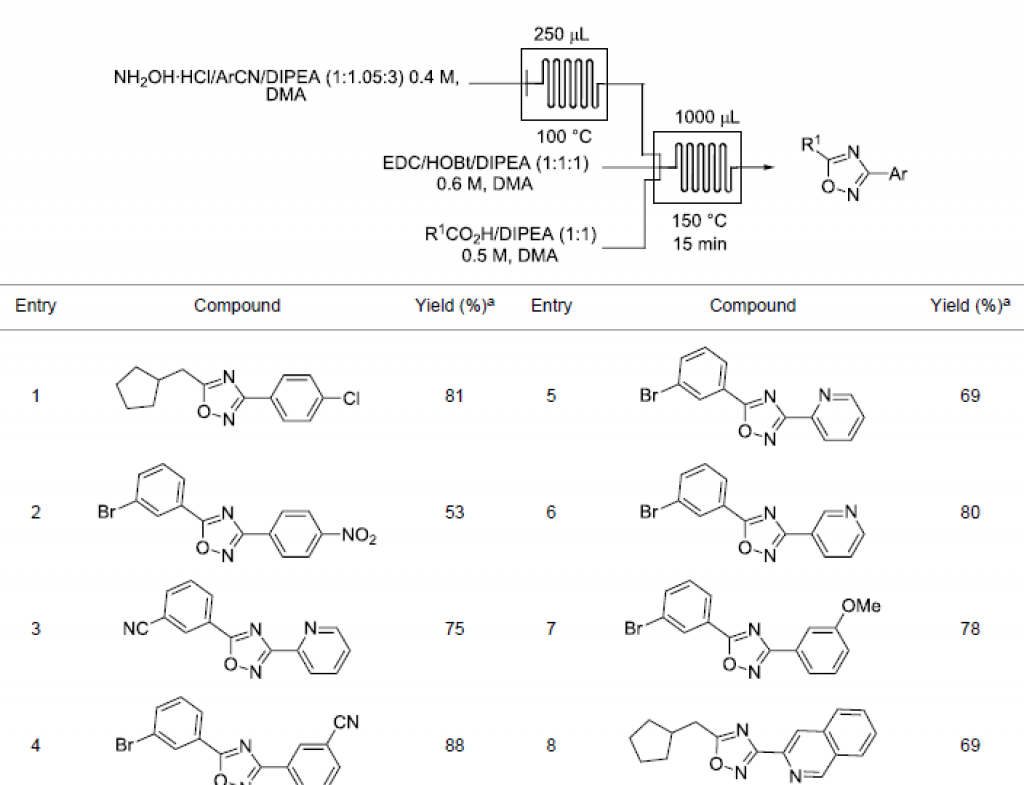
The synthesis of many analogous compounds for the purposes of testing is often referred to as library synthesis or high-throughput chemistry. Library synthesis is a crucial technique for rapidly exploring the chemical space of a molecule, allowing for the quick identification of lead compounds.
Traditionally, library synthesis is performed using traditional batch methods in multiple small flasks and vials, such as the Atlas HD Orbit system. However, with the advent of flow chemistry systems, many pharmaceutical research chemists switched to continuous flow chemistry for their library synthesis.
Conducting library synthesis using continuous flow chemistry techniques
Due to the benefits flow chemistry can offer chemists – faster reactions, higher selectivity, and ease of scale-up – it is an excellent method for the rapid synthesis of vast compound libraries and has been adopted in some form by all of the top pharmaceutical companies in the world. One flow chemistry reactor is able to automatically run 100s of experiments with minimal set-up time.
Find out more about conducting library synthesis in continuous flow
What are the 2 main types of drug synthesis process?
The pharmaceutical manufacturing domain encompasses two primary methodologies for producing drugs: chemical synthesis, which relies on chemical reactions, and bioprocessing, which harnesses the remarkable potential of microorganisms and cells to yield valuable compounds. Both approaches play integral roles in the drug synthesis process, leveraging distinct mechanisms to bring forth therapeutic substances.
The drug development process
Even at this early stage, scientists need to consider the final product and how it will be given to patients, and how easy it will be to develop and scale-up to manufacturing scale.
The use of crystallization in creating Active Pharmaceutical Ingredients (APIs)
Crystallization has long been used in chemical synthesis, as both a separation and purification step, and depending on the conditions used will provide crystals of different shapes and sizes.
Controlling the structure of the crystals formed is particularly important for the pharmaceutical industry, as the physical properties (shape, melting point, melting point etc.) of Active Pharmaceutical Ingredients (APIs) differ between different crystal structures, called polymorphs. For example, nanocrystals tend to increase the bioavailability of APIs due to their increased surface to volume ratio.
The size of the crystals produced can also greatly affect the physical properties of the bulk compound:
- Small crystals – improved packing density/flow of powders
- Large crystals – overcome filtration difficulties of small particles
Traditional methods of crystallization include; solvent evaporation, slow cooling and solvent/ vapor diffusion, and the crystallization process are typically monitored with a turbidity probe, due to ease of use and affordability. Discover a detailed breakdown of the crystallization process and its uses in industry here.
Sonocrystallization in Active Pharmaceutical Ingredient (API) development
Sonocrystallization has risen in popularity over traditional methods of crystallization due to polymorph control, reduced nucleation times, decrease metastable zone width (MSZW), reduced agglomeration and many more. Discover a detailed breakdown of sonocrystallization on the “what is sonocrystallization?” application page.
In recent years, alternative methods of monitoring crystallization processes (ATR-FTIR, FBRM etc.) have gained traction due to the additional information they can provide, such as chord length, particle size distribution, the presence of impurities, etc.
The Atlas HD Crystallization system is an automated jacketed reactor system, offering a turbidity probe and sonocrystallization module for complete control and measurement of the crystallization process.
Reaction calorimetry in drug development scale-up
Many chemical transformations release energy (exothermic reactions) or absorb energy (endothermic reactions), and chemists need to be particularly aware of these factors when seeking to scale up their chemistry to ensure sufficient safety protocols are in place.
Early identification of non-scalable reactions at the laboratory scale allows process chemists to select robust, safe, and scalable routes to drug candidates, saving development cost further through the process.
Reaction calorimetry can be performed for chemistry created in batch and flow techniques. Discover the reaction calorimetry application pages for an introduction to reaction calorimetry and its uses.
Absorption, distribution, metabolism, and excretion (ADME)
Before undertaking any in vivo trials, it is important to understand the absorption, distribution, metabolism, and excretion (ADME) of the drug molecule within an organism.
Drug compounds begin to break down as soon as they enter the body, with the majority of drug metabolism occurring in the liver facilitated by redox enzymes. The parent molecule is converted by these enzymes into new molecules called metabolites.
Researchers need to investigate the therapeutic properties of both the parent molecule and any metabolites produced. It can be incredibly difficult to synthesize metabolites from the parent molecule, as competing side reactions can occur, limiting the effectiveness of metabolite synthesis.
Electrochemistry has shown to be an effective method of selectively oxidizing key functional groups within a drug molecule, allowing researchers to directly access metabolites without the need for multi-step synthetic pathways. Electrochemistry modules effectively mimic the human liver, enabling chemists to easily test how their compounds will break down in the body.
Electrochemistry has been a traditionally difficult chemistry to perform, but modern advancements in flow chemistry have enabled easier access.
Utilizing the Asia Flow Chemistry Electrochemistry System for ADME testing
The Asia FLUX Electrochemistry module for the Asia Flow Chemistry System offers chemists the ability to mimic the human liver and rapidly test their compounds to see how they will break down in the human body. Performing electrochemistry in continuous flow offers far simpler and easier control over reactions conditions than traditional electrochemistry techniques, enabling drug discovery and development chemists to perform more experiments with greater accuracy in a shorter timeframe.
Drug discovery and development customer stories
A new dimension in drug discovery for Gedeon Richter
The Discovery Chemistry Department at Gedeon Richter in Budapest, Hungary, invested in an Asia flow chemistry system to aid researchers involved in the design and synthesis of original central nervous system (CNS) drugs, and enabled the researchers to access new chemistries previously impossible with traditional methods.
The system has extended the range of chemistries available to us, allowing us to work at much higher pressures and temperatures – sometimes above a solvent’s boiling point – to create completely new heterocyclic scaffolds."
Syrris products in drug discovery and development publications
Syrris products have been used in many drug discovery and development studies, including:
Preparative microfluidic electrosynthesis of drug metabolites
This paper describes the preparative electrochemical synthesis of phase I and phase II metabolites of a range of commercial drug molecules. In this study the Flux module is utilised to replicate hepatic oxidation, whilst maintaining high throughput at the preparative scale (100mg/hr) generating a range of phase I metabolites and Phase II Glutathione adducts on a range of commercially available drug molecules, therefore enabling full structural elucidation of these metabolites to be carried out by NMR (1D and 2D).
ACS Med. Chem. Lett. 2013, 4 (11), pp 1119–1123 | https://doi.org/10.1021/ml400316p
This publication uses an Asia flow chemistry system and the FLUX module
Synthesis of 4-Substituted Phthalazin-1(2H)-ones from 2-Acylbenzoic Acids: Controlling Hydrazine in a Pharmaceutical Intermediate through PAT-Guided Process Development
Using the Syrris Atlas HD Calorimetry system, researchers in the Process Development and Attribute Sciences groups at Amgen (US) have improved on a key process for controlling the levels of Hydrazine in a Pharmaceutical Intermediate characterized using Power Compensation Calorimetry (PCC).
Organic Process Research & Development 2015, 19 (7), pp 884-891 | https://doi.org/10.1021/acs.oprd.5b00135
This publication uses an Asia flow chemistry system
Discuss your pharmaceutical drug discovery and development chemistry with a Syrris chemist
Interested in how the Syrris range of flow chemistry systems, automated and manual jacketed reactors, reaction calorimeters, and scale-up reactors could help you discover and develop new and exciting chemistry? Speak to a member of the Chemistry team today.



