At Syrris, we believe that our existing customers are vital to our continued success. Creating and maintaining happiness with our customers across the world is the most important part of our business.
Our global customer support team is built on a foundation of scientific and product knowledge from passionate, motivated individuals. We assist our customers – new and old – with technical advice, troubleshooting, and product usage guidance. This ensures they get the absolute best from their Syrris products.
-
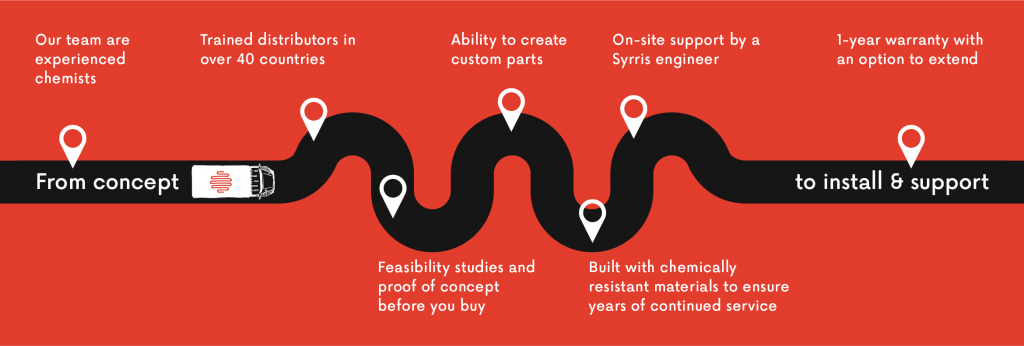
Global support team
Global Syrris support team, including 50+ distributors, gives you the help you need, when you need it, wherever you are
-
Product warranty
Get an extra 6 months added to your standard warranty cover (18 months total) free of charge when you register your Syrris product within 12 months of dispatch
-
Support from the experts
Friendly support team with a strong foundation of scientific and product knowledge